Abstract:
Celphos poisoning is a common occurrence in agricultural countries like India, particularly in rural areas. Its clinical manifestations vary depending on the amount of poison consumed and the time since ingestion, with a very high fatality rate. Currently, there is no specific antidote available for this poisoning. Management includes intensive monitoring along with immediate initiation of supportive care. Celphos poisoning is often associated with multisystem involvement and multi-organ failure in severe cases; however, haematological complications are uncommon. Here, we present a successfully treated case of a young male with accidental consumption of Celphos poison, who presented late to a tertiary health care centre. His clinical features were suggestive of haemolysis, and further evaluation revealed an underlying glucose-6-phosphate dehydrogenase (G6PD) deficiency.
Key words: Poisoning, G6PD Deficiency, Celphos, Aluminium Phosphide, Haemolysis.
Introduction
Celphos (aluminium phosphide) is a fumigant pesticide regularly used by farmers in agricultural countries for storage of cereal grains. Despite its highly toxic nature, it is widely used and an easily available pesticide. According to published data, its fatality rate varies from 40% to 80%.1 It is used in India under the names Celphos or Rice tablet. Each tablet weighs 3 grams, with a fatal dose ranging from 150 to 500 micrograms. After ingestion, it releases phosphine gas which causes non-competitive inhibition of the electron transport chain in cytochrome oxidase, leading to diffuse cellular hypoxia.2,3 Symptoms range from nausea, vomiting, palpitations, and abdominal discomfort in mild cases to severe organ failure in moderate and severe poisoning, including hepatic and renal failure with associated multisystem involvement.3,4 The time between consumption and death depends on the severity of poisoning.
Here, we present a case of a successfully treated 35-yearold male patient with Celphos poisoning and underlying glucose-6-phosphate dehydrogenase (G6PD) deficiency
Case report
A 35-year-old male patient with no significant past medical history was brought to the hospital with an alleged history of accidental consumption of Celphos on his farm earlier that morning. Within 6 hours, he developed breathlessness, yellowish discoloration of the eyes and abdominal pain. He was admitted to a government hospital, near his locality within three hours, where he received primary treatment and gastric lavage. Subsequently, his relatives brought him to the tertiary care centre on the third day.
On admission, the patient was afebrile, with a pulse rate of 88 beats/minute, blood pressure of 114/80 mmHg, respiratory rate of 40/minute, and oxygen saturation of 88% on room air. Clinical examination revealed pallor and icterus. On chest auscultation, bilateral crepitations were noted in the infrascapular regions. Abdominal examination showed distention with tenderness in the right hypochondriac region.
Arterial blood gas (ABG) analysis showed pH 7.425, partial pressure of carbon dioxide (pCO2) 31.2 mmHg, partial pressure of oxygen (pO2) 214 mmHg, bicarbonate (HCO3) 20 mmol/L, peripheral capillary oxygen saturation (SpO2) 89%, lactate 2.7 mmol/L, sodium (Na) 134 mEq/L, and potassium (K) 4.3 mEq/L. Due to SpO2 and PaO2 dissociation, methaemoglobin levels were checked, and which was 4%. The electrocardiogram (ECG) revealed sinus rhythm.
In view of breathlessness, oxygen support was initiated, and all the relevant investigations were done. Blood investigations showed anaemia (haemoglobin [Hb] 7.8 g/dL) and raised total leucocyte counts (26440/μL), while platelets were normal. Renal function tests showed serum creatinine of 0.3 mg/ dL, and serum urea 59 mg/dL. Liver function tests showed deranged values; total bilirubin 13.1 mg/dL (direct 2.2 mg/ dL), serum glutamic-oxaloacetic transaminase (SGOT) 134 U/L, and serum glutamate pyruvate transaminase (SGPT) 44 U/L. Prothrombin time was 13.6 seconds and international normalised ratio (INR) was 1.28. Glycosylated haemoglobin (HbA1c) was raised to 8.4%. The N-terminal Pro B-type Natriuretic Peptide (NT-proBNP) was also elevated at 328 pg/ mL. Serum creatine kinase-MB (CK-MB) was normal. His highresolution computed tomography (HRCT) chest scan revealed minimal left pleural effusion with basal atelectasis, and a few atelectatic changes in the right middle and lower lobes with mild ground glass opacities. His computed tomography (CT) abdomen showed fatty infiltration of liver, mild splenomegaly, and minimal peri-splenic fat stranding. His 2D-echocardiogram was normal with left ventricular ejection fraction of 60%.
Based on clinical findings and investigations, he was initially managed with intravenous antibiotics (meropenem), hepatoprotective drugs (ursodeoxycholic acid), antioxidants such as N-acetyl cysteine (NAC) infusion and glutathione, antipyretics, analgesics, multivitamins (vitamin C, pyridoxine) and intravenous fluids. Electrolyte imbalances were corrected as required. The patient’s urine was initially reddish coloured, resembling cola . Urine routine showed raised glucose and red blood cells (RBCs) (250/uL). Suspecting haemolysis, serum lactate dehydrogenase (LDH) levels were checked, and found to be elevated (2475 U/L). A peripheral smear showed predominantly macrocytic hypochromic RBCs with no haemoparasites. His G6PD levels were deficient at 4.2 U/g Hb. Direct Coombs' test was negative.
This rare case of Celphos poisoning presented with hyperbilirubinemia, methaemoglobinemia, and haemolysis. The patient improved with conservative treatment, recovered well and was discharged in a stable condition.
Serial investigations were conducted for monitoring(Table 1).
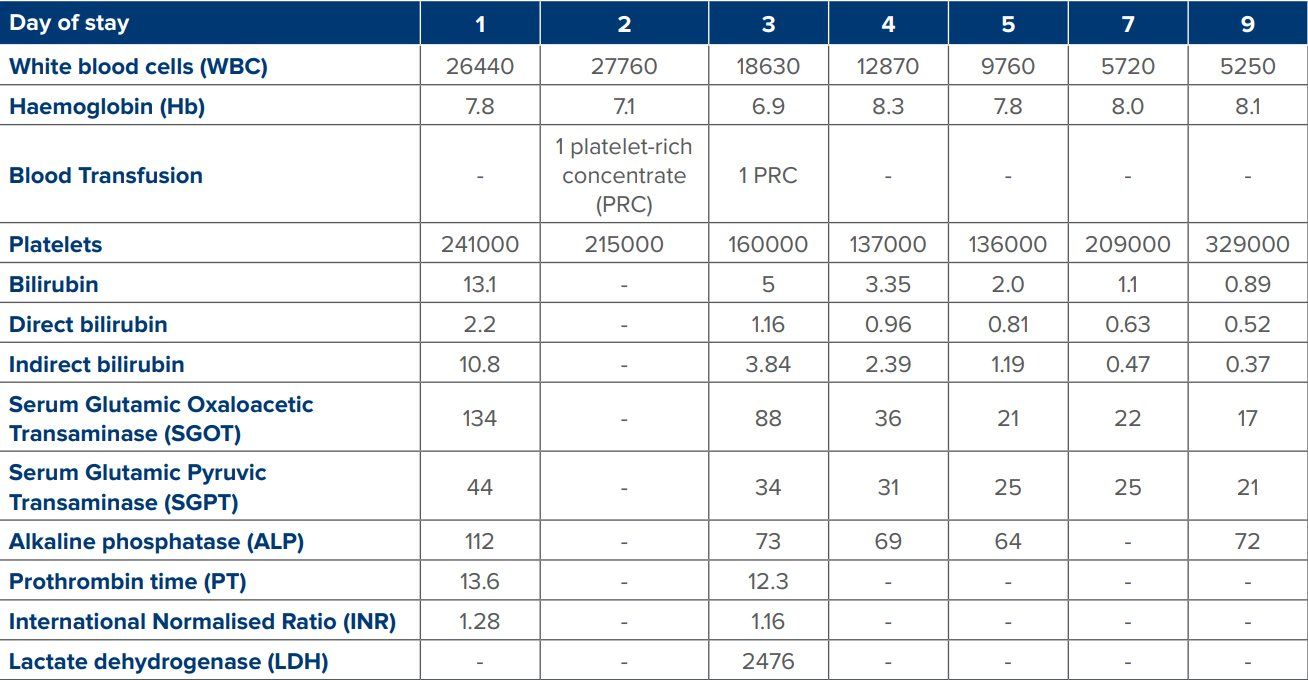
Figure 1: Showing serial investigations conducted for monitoring
Discussion
Glucose-6-phosphate dehydrogenase deficiency is a genetic disorder that affects red blood cells, causing their premature destruction. Glucose-6-phosphate dehydrogenase is an X-linked enzyme that acts as a catalyst in the hexose monophosphate (HMP) shunt pathway. In the HMP pathway, 6-phosphogluconolactone is produced from glucose-6- phosphate with the help of G6PD enzyme, generating nicotinamide adenine dinucleotide phosphate (NADPH). This NADPH is required to maintain reduced glutathione, which protects RBCs from oxidative damage. The most common clinical feature of this disorder is drug-induced haemolytic anaemia especially in adults.5
Many patients with G6PD deficiency are asymptomatic in the steady state. However, after exposure to trigger factors such as various medications, poisons, infections, fava beans, and metabolic abnormalities, haemolysis can occur causing anaemia, haematuria, and hemoglobinuria.6 In case of Celphos poisoning, haemolysis is the rare complication. In healthy individuals, after in vitro incubation of erythrocytes with phosphine gas, no haemolysis was found. The icterus in Celphos poisoning may be due to reduced liver perfusion causing direct damage or due to indirect hyperbilirubinemia, as in this case caused by haemolysis.7 On the other hand, Celphos may act as a trigger factor for haemolysis in G6PD-deficient individuals.
As we know, Celphos poisoning is usually fatal in most cases due to the lack of a specific antidote. In our case, the patient survived, and presented three days after ingestion with minimal left-sided pleural effusion, mild splenomegaly, indirect hyperbilirubinemia, mild methaemoglobinemia and reddish cola-coloured urine due to haemolysis. He was incidentally found to have G6PD deficiency.
After reviewing the literature, only 4 cases of G6PD deficiency with Celphos poisoning were reported,6,8-10 Surprisingly, all the patients survived, suggesting that G6PD deficiency might have a protective effect against Celphos poisoning. Oxidative stress and extra-mitochondrial release of free oxygen radicals have been suggested as a cause of aluminium phosphide toxicity. Due to the extensive RBC lysis in G6PD deficient patients, a theory has emerged that G6PD deficiency may prevent the disseminated oxidative stress caused by the phosphine-damaged RBCs.11 To investigate the protective effect of G6PD deficiency, research was done in rat hepatocytes, which demonstrated that G6PD deficiency significantly reduced hepatotoxicity of aluminium phosphide in rats.
Conclusion
As there is no specific antidote for Celphos, there are high mortality rates among patients. All possible alternative measures should be explored to save them.
The case described here indicates that a strong suspicion of G6PD deficiency should be considered in a case of haemolysis post Celphos poisoning. On the other hand, since all the reported cases of G6PD deficiency with Celphos poisoning survived unexpectedly, further research should be conducted to explore the potential for reducing fatality in Celphos poisoning by discovering agents that can temporarily induce G6PD deficiency.
The possible potential protective mechanisms include: reduced activation of toxic phosphine intermediates due to lower cellular activity, chronic adaptation to oxidative stress, enhancing alternative antioxidant defences, lower metabolic activity, reducing the impact of phosphine gas, attenuated inflammatory response due to altered reactive oxygen species (ROS), and altered or reduced cardiotoxic effects, particularly in myocardial cells.
Nitin Wadaskar, Prafulla Doifode, Rahul Arora. A Case of Celphos Poisoning with Glucose-6-Phosphate
Dehydrogenase (G6PD) Deficiency. Is There a Correlation? MMJ. 2025, March. Vol 1 (5).
References
- Mathai A, Bhanu MS. Acute aluminium phosphide poisoning: Can we predict mortality? Indian J Anaesth. 2010;54(4):302-7.
- Khosla SN, Handa R, Khosla P. Aluminium phosphide poisoning. Trop. Doct. 1992;22(4):155–157.
- Gurjar M, Baronia AK, Azim A, et al. Managing aluminum phosphide poisonings. J Emerg Trauma Shock. 2011;4(3):378- 84.
- Mukherjee MB, Colah RB, Martin S, et al. Glucose-6-phosphate dehydrogenase (G6PD) deficiency among tribal populations of India - Country scenario. Indian J Med Res. 2015;141(5):516-20.
- Farnaghi F, Owliaey H, Hassanian-Moghaddam H, et al. Intravascular Haemolysis due to Glucose -6- Phosphate Dehydrogenase Deficiency in a Patient with Aluminum Phosphide Poisoning. Indian Journal of Forensic Medicine & Toxicology. 2013;2:79-81.
- Soltaninejad K, Nelson LS, Khodakarim N, et al. Unusual complication of aluminum phosphide poisoning: Development of haemolysis and methemoglobinemia and its successful treatment. Indian J Crit Care Med. 2011;15(2):117-9.
- Humayun M, Haider I, Badshah A, et al. Protective role of G6PD deficiency in aluminium phosphide poisoning. J Coll Physicians Surg Pak. 2015;25(Suppl 1):S66-8.
- Srinivas R, Agarwal R, Jairam A, et al. Intravascular haemolysis due to glucose-6-phosphate dehydrogenase deficiency in a patient with aluminium phosphide poisoning. Emerg Med J. 2007;24:67-8.
- Vosooghi AA, Salmasi M. G6PD deficiency and aluminium phosphide poisoning. J Res Med Sci. 2018;23:83.
- Zamani N, Mehrpour O. Protective role of G6PD deficiency in poisoning by aluminium phosphide; are there possible new treatments? Eur Rev Med Pharmacol Sci. 2013;17:994-5.
- Salimi A, Paeezi M, Yousefsani BS, et al. Inhibition of glucose6-phosphate dehydrogenase protects hepatocytes from aluminium phosphide-induced toxicity. Pestic Biochem Physiol. 2017;143:141-6.

